Department of Infectious Diseases and Microbiology (IDM) faculty members have been awarded more than $150 million in funding from the National Institutes of Health (NIH) and other agencies over the past 10 years. Since 2003, Pitt Public Health has consistently ranked third nationally in NIH funding among comparable institutions. Our research involves students, postdoctoral fellows, and highly trained research staff.
The following are examples of standout funded IDM faculty research.
Complications of Antiretroviral Therapy
Assistant Professor Jeremy Martinson is leading research on genetic factors affecting cardiovascular complications of HIV therapy. The introduction of Highly Active Antiretroviral Therapy (HAART) has transformed the lives of many HIV-positive individuals. For some, however, HAART carries a risk of cardiovascular side effects, although the etiology of this is unknown. HIV infection is known to contribute to cardiovascular disease risk in some patients. Martinson and his team are investigating the extent to which host genetics, HIV infection, and treatment with HAART interact to give rise to cardiovascular disease in patients enrolled in the Multicenter AIDS Cohort Study. They are currently using a variety of high-throughput genomic and transcriptomic approaches to study this phenomenon.
HIV-1 Associated Neuropathogenesis and 3D Brain Organoid Models to Study Neurodegenerative Diseases and Therapeutic Strategies to Mitigate Chronic Inflammation
HIV-associated neurocognitive impairment (HAND) is commonly observed in AIDS patients worldwide. During early infection, HIV-1 enters the Central Nervous System (CNS) and resides in macrophages/microglia and causes neuronal degeneration and inflammation in the brain. HAART therapy has improved the quality of life and longevity of HIV-1 infected patients; however, with aging, the prevalence of HAND is high in this population. Velpandi Ayyavoo’s laboratory focuses on understanding how HIV-1 induces neuronal dysfunction and destruction and how to detect them during the early stages as neurodegeneration occurs over a long period of time resulting in clinical manifestations. However, studying brain pathologies are complicated due to the lack of physiologically relevant models with encompasses the cell lineages including neurons, astrocytes and glia. Thus, her laboratory has developed a 3D-brain organoid model that will enable studies to delineate neurodegenerative diseases (Reis et al. 2020, Scientific Reports). Using this model her group is focusing on addressing how HIV infection induces chronic inflammation in the CNS and the consequences of inflammation on neurons and other bystander cells in the brain. Additionally, research is focused on developing immunotherapeutic treatment strategies that will reduce infection induced inflammation by overexpressing microRNAs that could potentially block inflammatory factors. The therapeutic potential of these miR-microglia will be assessed in the 3D-organoid model and in humanized mouse model.
Modeling HIV Reservoir, Lymphoid Tissue Pathology and Human Immunology in vivo
Recent studies in the laboratory of Assistant Professor Moses Bility have developed rodent models with human immune cells along with primary and secondary human lymphoid tissues for studying the HIV reservoir and human immunology. These models are being used to delineate the role and mechanisms of myeloid cells-T cells interactions in live-attenuated HIV-induced immunity in human secondary lymphoid tissues. These models are also being used to explore the mechanisms by which myeloid cell polarization facilitate HIV persistence, promote chronic inflammation, and modulate lymphoid fibrosis. Additionally, in collaboration with others, these novel rodent models with human immune system, including humoral immune system, are being use as a platform to rapidly develop human therapeutic antibodies against various pathogens.
AIDS Education and Prevention
“Disparities in health care, the stigma of HIV/AIDS, and the need for more timely testing and treatment are still barriers to delivering and improving HIV care,” said Linda Frank, Associate Professor. Frank is a principal investigator at the Pennsylvania/MidAtlantic AIDS Education and Training Center, headquartered at Pitt Public Health since 1988. The center is a national leader in supporting the training and education of health professionals who care for the thousands of people infected annually with HIV in the United States. A recent grant will allow scientists like Frank to continue to provide critical training to health professionals to reduce barriers to HIV care, increase HIV testing, integrate HIV prevention into primary care, and improve linkages to HIV treatment and expert clinical management of HIV and other comorbid conditions, such as hepatitis, sexually transmitted infections, tuberculosis, and substance use.
Herpesviruses
Recent studies in the laboratory of Assistant Professor Giovanna Rappocciolo, have identified DC-SIGN and langerin, two C-type lectins, as the main receptor used by human herpesvirus 8 (HHV-8), or KSHV, the cause of Kaposi's sarcoma, to infect dendritic cells, dermal dendritic cells, B lymphocytes andmacrophages. These findings established the first in vitro model of productive HHV8 infection of its natural target cells.
HIV-1 Immunopathology
Dr. Rappocciolo's focus has been the study of the relationship between antigen presenting cells mediated HIV trans infection and HIV-1 disease progression. Strikingly, dendritic cells, B cells and macrophages from patients able to naturally control HIV-1 disease progression, called controllers, do not transfer HIV-1 to bystander CD4T cells and this defect seems to be related to lower contents of cellular cholesterol. Currently, Dr. Rappocciolo's lab is working on the identification of soluble biomarkers of HIV disease progression as related to the modulation of cell cholesterol content. More recently, the work on the role of trans infection in the regulation of HIV disease has been expanded to define the importance of B cell mediated trans infection in the establishment of the HIV latent reservoir in a subsets of CD4T cells, the naive CD4 T cells. Her lab is now expanding on this seminal study to understand the interactions between B cells and CD4T cells subsets that lead to HIV transfer and the establishment of the latent reservoir.
Tuberculosis
Nearly one third of the world’s population is infected with Mycobacterium tuberculosis, the bacterium that causes tuberculosis (TB). Although the vast majority of M. tuberculosis-infected people never experience the clinical symptoms of TB, there are almost 11 million new cases of TB and two million TB-related deaths annually. HIV is strongly associated with active TB, even in individuals with well-preserved CD4+ T cell levels, and TB is the leading cause of death in HIV-infected individuals. The causes of active TB are poorly understood, but are likely to include changes in anti-mycobacterial immune responses in granulomas, the site of M. tuberculosis infection. Assistant Professor, Josh Mattila and his lab are focused on understanding the immune environment in lungs and TB granulomas. His particular interest is examining interactions between myeloid cells, including macrophages and neutrophils, and T cells in this environment with the objective of identifying factors that differentiate pathologic and protective responses.
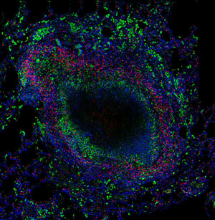 T cells (red) and macrophages (green) are present in distinct regions in granulomas where they cooperate to control Mycobacterium tuberculosis infection.
T cells (red) and macrophages (green) are present in distinct regions in granulomas where they cooperate to control Mycobacterium tuberculosis infection.
Emerging Infections
Influenza virus is a respiratory pathogen that targets the lung, and innate immune responses serve a key role in providing early antiviral immunity and limiting disease in acute infection. However, the relationship between dendritic cells and immunity in the lung is not well defined. Professor Simon M. Barratt-Boyes and his team have used the murine model to examine the impact of plasmacytoid dendritic cells in the early immune response to influenza virus infection. They are also studying the dynamics of the dendritic cell response to avian influenza virus infection in the lung of nonhuman primates, which they believe serves as a more robust model of influenza in humans than do mice.
HIV Prevention and Planning
The HIV Prevention and Care Project (HPCP) is one of the longest running technical support and HIV Planning programs for any statewide jurisdiction in the United States. Founded in 1992, the program has generated more than $40 million dollars in state and federal pass through funding to engage and empower people living with HIV, organizational stakeholders, and communities impacted by or at-risk from HIV. With a staff of more than 15 scientists, trainers, stakeholder engagement specialists, and technical experts, the HIV Prevention and Care Project collaborates with the Pennsylvania Department of Health’s Division of HIV Disease and related state agencies to provide organizing support, meeting facilitation, scientific expertise, intervention delivery, community advisory board (CAB) development and maintenance, pilot programs and community-based research for some of Pennsylvania’s most vulnerable communities, and capacity building and technical assistance for the Commonwealth of Pennsylvania.
The mission of the HIV Prevention and Care Project is to facilitate HIV planning in Pennsylvania and support scientifically-based interventions to reduce HIV infection rates and lower morbidity and mortality associated with HIV and AIDS. We envision an AIDS-free future where essential HIV prevention and treatment services are accessible to all who need it and where most-at-risk communities are structurally empowered in state and local public health strategy development and implementation to eliminate HIV-associated morbidity and mortality. The project’s models for achieving integrated HIV prevention and care planning and for structural, meaningful involvement of most-at-risk communities in these processes have been cited as best-practices by federal entities including the Health Services and Resources Administration (HRSA) and NASTAD. Dr. Mackey Friedman serves as PI and Drs. Sarah Krier and David Givens serve as Co-PIs.
STD-to-PrEP (2019 - ) is a demonstration project focused on understanding a sexual health navigation intervention on HIV-negative individuals’ HIV prevention decision-making, access to oral PrEP (pre-exposure prophylaxis), and associated care continuum (e.g., PrEP uptake, adherence, and maintenance). Demonstration project participation includes sexual health navigation services and associated PrEP experience follow-up. This project is funded by the Centers for Disease Control and Prevention (CDC) and conducted in collaboration with the Pennsylvania Department of Health and the Allegheny County Health Department. The project is led by Dr. Sarah Krier (mPI), Dr. Mackey Friedman (mPI), Dr. Teagen O’Malley (Project Director), and Harley Roth (Project Manager). Project findings are forthcoming and will be shared with key stakeholders to support enhanced sexual health navigation and PrEP access and care in Allegheny County, as well as the larger public health community.